The story of the most epic pset ever by Laura N. '09
If you correct my problem set in the comments, I will get angry. And you won't like me when I'm angry.
This semester I’m taking a class called “Sustainable Energy,” and so far, it’s pretty awesome. It’s one of the few classes where I really feel like I’m learning a lot. I mean, I’m sure I learned a lot in 2.005 but it’s hard to feel like you’re learning, you know? You try doing a problem that requires you to use the heat equation and see if you finish feeling less confused than you started.
But in Sustainable Energy we learn lots of actual, concrete stuff in every lecture. So every Tuesday and Thursday I leave class thinking, “So that’s how carbon sequestration works,” and feeling all enlightened and such. Part of this is because the class is HUGE in scope. We tackle the energy problem from what seems like every possible angle- economics, engineering, science, politics, nuclear, mechanical, physical, electrical, you name it.
To illustrate this, I documented some of the sleepless night I spent the other day working on what was quite possibly the most epic pset of all time.
It started out with a pair of daunting circuit problems which I probably would have breezed through back in the Stone Age when I was taking 8.02 (seriously, I feel so old now it’s not even funny) but were definitely beyond the rusty remains of my E&M skills.

After awhile I visited the handy “Conner 2 library” (fancy name for a bookcase in the floor lounge where people stash old textbooks they don’t want or need anymore for anyone on the floor to use) and grabbed a copy of the 8.02 course notes. What’s impedance again? And inductance…is that thing…it’s like the opposite of capacitance, so they get all wavy in an RLC circuit. (Believe it or not, freshman year I actually went back to my high school and successfully taught the lesson in RLC circuits to the seniors in AP Physics. I must be getting senile in my old age.) The textbook was not very helpful.
I eventually had to give up on the circuit problem for a little while, even if just to give my mind a break. Problem 2 was about carbon dioxide emissions. If there are 0.9 hydrogen atoms per one carbon atom in every molecule of coal, and 2 oxygen atoms per one carbon atom in carbon dioxide (well, duh) then how much CO2 is produced by burning one kilogram of coal? I had a feeling this was some kind of chemistry problem, but I shoved that thought to the dusty back corner of my mind, right next to the word “stoichiometry.” I just made up some ratios that made sense and went on with my life.



Me: “But…but…why can’t you just do it the way I did, with ratios?”
Adelaide: “Well, you can, this just makes it easier.”
Me: “…okay this is why I’m a physics person.”
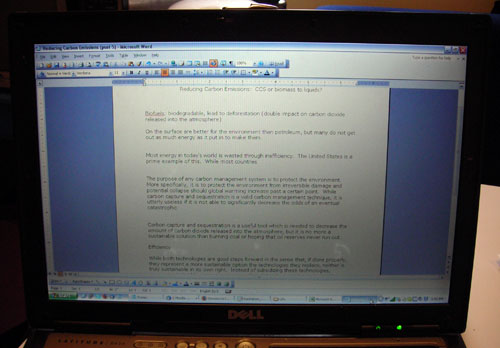
Alright, time for Problem 4. Problem 4 was an essay. I’m not kidding! Write two pages about fuels derived from biomass and carbon sequestration and whether you think these are sustainable technologies. This pset has everything. Except like, some nuclear problem.

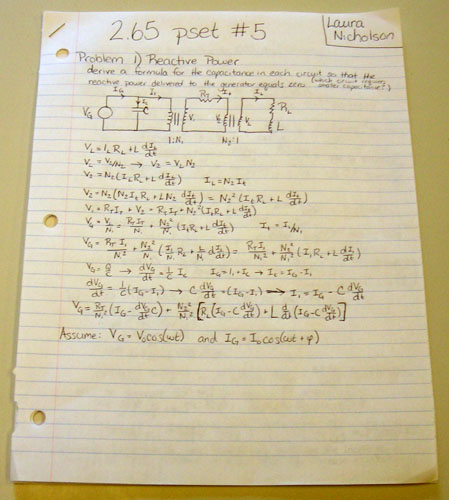
today only I did the EMI and AC chapter from physics and that RLC circuit is fun
First! Haha!
i could have helped with the chem question btw i still know it!
YAY, MIT PSETS FTW!
STOP POSTING PORNO YOU CREEPS!!!
A two page essay in addition to a bunch of undecipherable equations to solve for? Only at MIT :D.
This most definitely sounds like a class that I would enjoy being in….
this isn’t really about the blog, but…
who do i contact about missing pieces of my application? one of my teacher recommendations was sent like a month ago, and its still not showing up in my application tracking thing as being processed. is this something i should worry about?
@anon 6:54: Try Kim Hunter. She has a blog on this site, or you can try e-mailing her directly.
I agree with Mohit too!! MIT classes seem so interesting!!
I decided a few months ago that I was interested in applying to MIT, and found this site. While here, I read this really cool, funny blog about sarcasm (and the lack thereof) in Spain. Now, coming back today, I read through this and your Halloween blog, which made me think of the sarcasm one, and I was like, “Ha, wouldn’t it be funny if it was the same girl?” And IT WAS!!
The whole point of this was that I wanted to tell you that your blogs are SO funny and interesting, I’m not a big blog/internet journal reader, but I love reading yours! =]
Agree with Mohit !
And I agree with Dhvanit!!
And I agree with Dhvanit!!
<3 chemistry
<3 chemistry
~heart~ chemistry
(what the above message was supposed to say)
I suppose you are doing Physics and Chemistry. Its FUN.
Hey this really sounds intresting and real.I just finished high school not not more than six months ago but some ideas are alredy fying away….but thanks to my high school chem textbook.
What’s course six? Your codes are so confusing